Coral propagation at Plantages PortoMari-8 Months
By Maureen Kuenen mkuenen@cura.net & Todd R. Barber reefball@reefball.com
Obserations
Propagation, especially by volunteers, can weaken corals and make them vulnerable to several dangers.
In our first propagation we made several mistakes including poor water quality in our propagation tank due to a pump failure, handling corals without latex gloves, and transplanting the corals before the summer algae bloom. Even so, we achieved excellent survival rates in most species and good survival rates in the more sensitive species.
One of the problems we experienced was that algae started to
(Below: Summer Algae Bloom 2 months after the first
deployments)
grow

on damaged parts and from this start it
could expand to cover healthy tissue.
This covering can cause food starvation and light reduction weakening
the coral even more. Algae can also attract coral predators; presumably
parrotfish that not only scrape the algae of but also can damage large parts of
healthy coral tissue doing so.
Our experience with specific local species:
 Porites
porites and Madracis mirabilis (finger and pencil
coral) showed recovery (regeneration of damaged tissue) of such predator
actions can easily occur within a month as long as there is enough living
tissue left and no further impact on the coral fragments occurs. This may
indicate that predators can destroy coral fragments but also rescue them from
coverage by algae. Such relationships have been noted in the Pacific of
Butterfly fish eating coral eating nudibranchs helping to save them rather than
being the predator it was always assumed.
Porites
porites and Madracis mirabilis (finger and pencil
coral) showed recovery (regeneration of damaged tissue) of such predator
actions can easily occur within a month as long as there is enough living
tissue left and no further impact on the coral fragments occurs. This may
indicate that predators can destroy coral fragments but also rescue them from
coverage by algae. Such relationships have been noted in the Pacific of
Butterfly fish eating coral eating nudibranchs helping to save them rather than
being the predator it was always assumed.
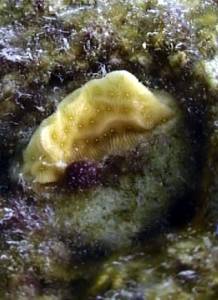
Agaricia agaricites (Lettuce Coral) has adaptation problems in
the beginning that are shown by bleaching. However as long as they hold, don't
show damaged tissue and don't get too much "attacks" from algae they
show a very healthy appearance after 4 to 5 months. Algae can block outgrow at
the base but when the algae are gone clear outgrow has been observed.
Eusmilia fastigiata (Smooth Flower Coral) seems to withstand algae growth very well and after a short period of showing signs of stress (bleaching), damaged tissues heal very fast and the fragments look good.
High survival of corals with slow growth rates like Montastrea annularis. (Boulder Star Coral)
Madracis and Acropora (Finger and Staghorn Corals) are very fragile and easily damaged (mechanical damage e.g. kicking of fins). Madracis recovers very fast. Growth tips of Acropora as well.
Branching corals are very susceptible for breakage and therefor developed the capability to recover fast. New tissue grows on broken parts and new colonies originate this way. This capacity of fast asexual reproduction highly contributes to recovery of damaged reefs. Because recovery starts right away, there is less chance that substrates will be overgrown by other benthic species (High Smith 1982 uit Brown and Haward)
Below: Acropora when new, 2 months and 8 months old



Gorgonians: The results vary per individual. They usually grow at very fast but some develop a strong base and others don't. Also some grow fast quickly branching and others grow slow and just have two branches. They easily form new branches after breakage.

Monitoring
Program:
To monitor an overall impression of the corals, you must look at mortality since growth rates don't always give impression of health. Damaged corals need energy for recovery and won't grow as fast while it can be perfectly healthy. Growth rates are also slow the first 2-4 months for many species as they divert all energy to “basing” rather than upward growth when first propagated or transplanted.

Picture of Reef Balls
at 2 months.
Details
Status coral plugs July 3, 2001 (90 days)
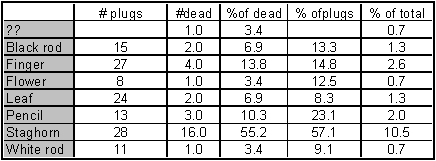
In total 19.08 % of the plugs died
Order of % of dead:
Staghorn, Finger, Pencil, Leaf/black rod, white rod/??/flower
Order of % dead of total plugs (placed fragments thus relatively)
Staghorn, Pencil, Finger, Black rod, Flower, White rod, Leaf.
That makes the Staghorn most vulnerable followed by the 2 other branching corals Pencil and Flower.
They also have powerful regeneration capability so more careful treatment may give better results.
(See www.artificialreefs.org à Scientific Resources à Coral Propagation and Transplant Notes for more careful treatment recommendations by coral species)
Although we transplanted maze coral/blushing star coral/star coral, brain corals, mustard hill coral.
There are not enough plugs made of these species so no real
conclusions can be drawn. However, here is the raw data.
Monitoring interval: March-October
subject # dead % dead # alive % alive Total # plugs Mortality
total coral
plugs 43 28 112 72 155
Black sea rod 2 15.4 13 84.6 15 low
Blushing star coral 0 0 1 100 1 not enough samples
Brain coral 0 0 5 100 5 possibly low
Finger coral 13 54.2 16 45.8 24 high
Flower coral 1 12.5 7 87.5 8 low
Leaf coral (lettuce) 0 0 25 100 25 very low
Maze coral 1 16.6 5 83.4 6 possibly low
Mustard hill coral 0 0 1 100 1 not enough samples
Pencil coral 8 53.3 7 46.7 15 high
Purple searod 0 0 6 100 6 possibly low
Staghorn coral 17 60.7 11 39.3 28 high
Star coral 0 0 3 100 3 not enough samples
Starlet coral 0 0 2 100 2 not enough samples
White searod 1 9.1 10 90.9 11 low
There is a total survival of 72% that
is not so bad after the heavy algae growth from March till about August. Leaf coral (Agaricia agaricites) seems to
survive best. Even the plugs that were considered dead before "came
back" and started to grow again after most of the algae disappeared. The
disappearance of the algae went simultaneously with high recruitment of snails
(Cerithium sp.). Also for other species like Porites porites (finger coral), Acropora
cervicornis (staghorn coral) and Madracis mirabilis (pencil coral) a clear
outgrow of tissue was observed over the base of the cement plugs were algae
were removed. We actually removed lots of the algae manually and after that we
believe no new outbreaks of algae occurred and also the snails started to keep
the plugs clean. It might be a combination of circumstances of manual removal,
disappearance of certain algae and the recruitment of the ceriths that created
much better opportunities for the coral plugs. Gathering local cerths and concentrating
them on the plugs might be a possible intervention where algae blooms are occurring.
Although the Acropora c. still has
the lowest survival rate of 39.3%, most of the plugs that are left over are
doing extremely well with many new branches. In 2 cases a tip or branch broke
off but the recovery was fast and even more new grow tips developed (like with
some plants that you have to prune to make them grow better). These plugs could
therefore function as donor colonies for new coral plugs in the future so no
fragments have to be taken from the wild. Our experience with this species in
this project and other is that under no circumstances you should touch this
coral with your hands (wear latex gloves). They seem to be extra vulnerable for
contact with human tissues while breakage (quite an impact) doesn't affect them
so much. Rapid Tissue Necrosis (RTN) is
also common with Acropora in the aquarium trade…it is associated with stress,
low water quality and shock. RTN looks like bleaching, but careful examination
will show a sluffing off of clear or
white dead tissue starting at one point on the coral and rapidly advancing.
often in hours or a few days. When this
occurs, the only cure is to re-fragment the coral above the dead tissue and
then re-plug it.
Also Porites p. and Madracis m. have
a high mortality rate but I have seen no other species trying to recover so
hard as these ones. Specially Porites suffered a lot from predation, large
parts were scraped off but when there was enough tissue left over the tissue
grew back very fast. Most plugs that are left over now start to cover the base
of the plug or make new branches. New tissues are whitish so do not be confused
with bleaching. Madricis also suffered some predation but because of the small
size of the fragments the main problem we experienced was the algae. Now that
the algae are gone, they have the
chance to show how well they will do (we are not certain at this stage yet).
The Montastrea a. is a very strong coral
and can withstand a lot of stress. This was mainly observed with the new coral plugs,
where grow out over the plug is clearly observed. Because this species is (like
Agaricia a.) a main reef builder it should be used for future plugs in spite of
it's slow growth rate.
Agaricia a likes darker spots as it
is often a deeper water species. In the beginning lots of algae were growing on
the upper surface that died off while the lower surface stayed alive. Specially
the smaller fragments had a hard time to fight the algae and it looked as if
the weren't going to make it. Now most of the plugs are recovering and especially
at the sides with the least amount of direct sunshine the plugs grow best.
The Eusmilia f. plugs grow slowly but
surely. They don't seem to be bothered by the algae so much. Probably because
they have big polyps.
The gorgonians seemed to do very well
in the beginning but now show some problems. Especially some white sea rods show
signs of broken tips, dried up tissue and thin branches. Also a few black sea
rods show thin branches. These changes in morphology may reflect transplant
distances, lighting difference or even local planktonic differences.
All brain coral do well. They are fairly
strong can stand high stress levels for a short while but again they are slow
growing. Other species need more
samples to draw any conclusions.
Before the coral spawning (September/October)
a few Reef Balls were brushed off to remove excess of algae. In November coral recruits were observed with special
blue light (since they are fluorescent ). In December they could be observed
with the naked eye. All recruits were found on the cleaned Reef Balls and most
on the Ultra Ball. There are 2 species observed so far the identification is
not clear yet (Madracis and Eusmilia?).
Some statements from literature:
In studies of Connell (1973) and Bak and Engel (1979) mortality
of juvenile corals on reef unaffected by human disturbance is was resp. 36% and
32% during 11 and 6 months study periods. The causes of mortality were
sedimentation, competition with coralline algae on shallow reefs and possible random grazing and/or predation by
parrot fishes.
Sedimentation: Decreased light values/increased energy
consumption and possible reduction in planktonic food. (Bak '78)
Bacterial infections are often the result of corals
protecting themselves against outside stresses by mucus secretion. This may
contribute to increase of the blue green algae that are responsible for the
black band disease. That means mucus is a protection but also weakens the
coral.
Harold Hudson 1981: Tolerance for transplantation:
Montastrea annularis from deeper water to shallow water showed reduced growth rates and severe
mortality while transplanting from inshore to off shore showed only slightly
reduced growth rates ( increased environmental tolerances).
Bak, R. P. M. (1978). “Lethal and sublethal
effects of dredging on reef corals.” Mar Pollut Bull 9(1): 14-16.
Bak, R. P. M. and M. S.
Engel (1979). “Distribution, abundance, and survival of juvenile hermatypic
corals (Scleractinia) and the importance of life history strategies in the
parent coral community.” Marine Biology 54: 341-352.
Connell, J. H. (1973).
Population ecology of reef-building corals. Biology and Geology of Coral
Reefs Volume II: Biology 1. O. A. a. R. E. Jones. New York, Academic Press: 205-245.
Hudson, J. H. (1981).
“Response of Montastrea annularis to environmental change in the Florida Keys.”
Proc 4th Int Coral Reef Symp, Manila 2: 233-240.